Authentic Sarapin™ is defined by several characteristics: it is a sterile distillate made from fresh Sarracenia purpurea using the original, validated manufacturing process from High Chemical when the formulation and manufacturing process was initially approved. Products made from dried botanical powders are not true Sarapin™ and do not meet the scientific, historical, or manufacturing standards that define the formulation.
Why does this matter? Because in clinical and research environments, the identity and consistency of a product determine whether it can be trusted by physicians. When the starting material changes, the process changes, and the resulting preparation is no longer the same substance—no matter what it’s called. Authenticity directly affects quality, reproducibility, and professional confidence, especially for clinicians who rely on predictable characteristics and procurement teams who must ensure compliant sourcing.
USSF is the sole manufacturer of real Sarapin™ and the only provider of Sarapin™ API. This blog clarifies what authentic Sarapin™ is, how it is uniquely produced, and why substitutes made from dried leaf powder are not equivalent.
Understanding Real Sarapin™: The Core Facts
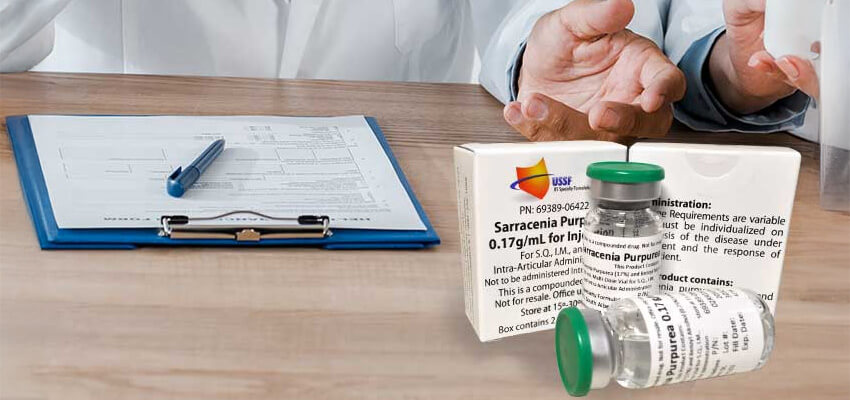
Real Sarapin™ is a sterile, cGMP‑manufactured distillate made exclusively from fresh Sarracenia purpurea plants—not dried leaf powder or ethanol extractions of a specific part of the plant. Only USSF produces authentic Sarapin™ also known as Pitcher Plant Distillate, and USSF is also the sole provider of Sarapin™ API for pharmaceutical and clinical manufacturing. This API has been historically used for treatment in humans as well as equine (horse) veterinary applications. Other products made from dried botanical powders are not true Sarapin™ and do not meet the scientific, historical, or manufacturing standards of the original formulation.
What Is the Real Sarapin™
Sarapin™ is a botanical‑derived injectable preparation historically used in pain management procedures such as trigger point, prolotherapy injections and acupuncture. Its defining characteristic is its origin from fresh whole Sarracenia purpurea, a North American carnivorous plant.
Authentic Sarapin™ is produced through a controlled distillation process, yielding a sterile, standardized solution that aligns with the traditional formulation clinicians have expected over the past 120 years.
Real Sarapin™ is not a generic plant extract, nor is it interchangeable with products made from other dried plant parts or powdered extracts. Its identity is tied directly to:
- Fresh plant inpu
- Precise distillation methods
- Controlled cGMP manufacturing
- Consistent chemical profile and quality attributes
This is what distinguishes true Sarapin™ from substitutes.
Interested in Sarapin? Discover detailed product information and clinical applications.
Learn More About SarapinHow Authentic Sarapin™ Is Manufactured— Fresh Sarracenia purpurea, Not Dried Leaf Powder
The defining scientific and manufacturing difference is simple and critical:
Authentic Sarapin™ = Fresh Sarracenia purpurea → Sterile Distillate
Imitation Products = Dried Leaf Powder → Crude Extract
USSF follows the original, validated approach from High Chemical:
- Fresh Sarracenia purpurea plants are harvested and processed immediately.
- The plant material undergoes a controlled distillation, not maceration or solvent extraction.
- The resulting distillate is sterile‑filtered, standardized, and packaged all under CGMP manufacturing conditions.
- Every batch is produced in a regulated facility with full documentation and quality controls.
This process preserves the characteristics that define Sarapin™ as one of the oldest and unique botanical preparations.
Why Powder‑Based Products Are Not True Sarapin™
Some companies—primarily in the veterinary market—sell products labeled as “Sarapin™” but their starting material and handling are not consistent with the original Sarapin™ recipe. This is not equivalent for several reasons:
- The plant species may not be the same.
- The harvesting and drying processes are not the same.
- Solvent based extraction methods extract a different chemical profile than the USSF method.
- Powder‑based extracts do not match the composition of a fresh‑plant distillate.
- These products do not follow the historical or scientific definition of Sarapin™.
- They are not manufactured to USSF’s CGMP standards, based upon process transfer from High Chemical the original manufacturer and decades of experience.
For clinicians, researchers, and procurement teams, this distinction is essential: the starting material, the controlled distillation, and the CGMP manufacturing process determines the identity of the final product.
Why Other Products on the Market Are Not True Sarapin™
Despite similar naming, powder‑based veterinary products:
- Do not use fresh Sarracenia purpurea
- Do not follow the validated distillation method
- Do not meet the identity criteria of authentic Sarapin™
- Do not originate from the original manufacturing source
- Do not supply an API suitable for pharmaceutical use
These products may be marketed as “Sarapin™,” but they are not Sarapin™ in the scientific, regulatory, or historical sense.
USSF: The Sole Manufacturer of Authentic Sarapin™
USSF is the only organization that:
- Manufactures real Sarapin™ from fresh Sarracenia purpurea
- Produces Sarapin™ under cGMP
- Maintains the original, validated distillation process
- Supplies Sarapin™ for clinical use
- Provides the Sarapin™ API for pharmaceutical and manufacturing partners
Real Sarapin™ API Availability
In addition to finished product, USSF is the exclusive provider of Sarapin™ API, enabling:
- Contract manufacturing
- Research and development
- Formulation studies
- Regulatory submissions
- International distribution partnerships
This API offering is a major differentiator, and a key reason USSF is the central authority for all Sarapin™‑related sourcing.
Real Sarapin™ vs. Powder‑Based Extracts
Real Sarapin™ vs Powder-Based Extracts Comparison Table
| Criteria | Authentic Sarapin™ (USSF) | Powder‑Based Alternatives |
|---|---|---|
| Source Material | Fresh Sarracenia purpurea | Leaf Powder |
| Manufacturing Method | Controlled distillation | Chemical solvent extraction |
| Regulatory Standard | CGMP | Varies; often non‑GMP |
| Identity | True Sarapin™ | Not Sarapin™ |
| Clinical Consistency | Standardized | Variable |
| API Availability | Yes – USSF only | No – often not approved API manufacturer |
Why Sarapin™ Authenticity Matters
For clinicians and procurement teams, authenticity affects:
- Consistency
- Regulatory alignment
- Clinical confidence
- Supply chain reliability
- Research reproducibility
Using nonequivalent products introduces variability that can affect clinical protocols and procurement compliance.
How to Verify Real Sarapin™
Confirm that the product is manufactured by USSF
USSF is the only manufacturer of authentic Sarapin™ for Injection and the only provider of Sarapin™ API. If the product does not originate from USSF, it is not real Sarapin™.
Supporting identity criteria (all of which only USSF meets):
- Manufactured from fresh Sarracenia purpurea
- Produced as a distillate, not a powder‑based extract
- Made under cGMP conditions
- Backed by the availability of Sarapin™ API (exclusive to USSF)
If a product fails the first criterion – USSF as the manufacturer – it cannot be authentic Sarapin™. The additional criteria simply explain why USSF is the only source: because no other manufacturer uses fresh plant material, follows the validated distillation process, or produces Sarapin™ under cGMP with an API offering.
FAQ
What is real Sarapin™?
A sterile distillate made from fresh Sarracenia purpurea, manufactured exclusively by USSF.
Why is fresh plant material important?
Fresh plants preserve the chemical profile required for authentic Sarapin™; solvent extracts which are dried to powders do not have the same chemistry.
Are veterinary products labeled “Sarapin™” the same?
No. Powder‑based extracts are not true Sarapin™ and do not follow the validated manufacturing process.
Who manufactures Sarapin™ today?
Only USSF.
Does USSF supply Sarapin™ API?
Yes. USSF is the sole provider of Sarapin™ API.
Conclusion
USSF remains the only manufacturer of authentic Sarapin™ and the exclusive source of Sarapin™ API. By maintaining the original distillation process (transferred from High Chemical) coupled with CGMP standards, USSF provides clinicians, researchers, and procurement teams with the clarity, consistency, and authority the market has lacked.
Tags: real sarapin, sarapin, sarapin injection, sarapin injection cost, sarapin injection price, sarapin injections