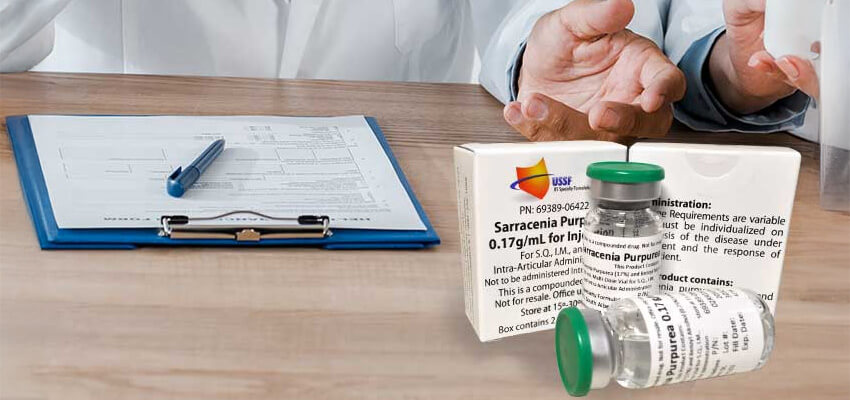
Botanical preparations have appeared in professional healthcare discussions for decades, particularly in fields that involve musculoskeletal care and injection based procedures. One such preparation is Sarapin, a plant derived injectable historically sourced from Sarracenia purpurea (pitcher plant).
This overview provides general educational information about Sarapin’s background, manufacturing considerations, and distribution policies. It does not describe clinical uses, safety, or efficacy and is not intended as medical advice.
Botanical Background and Historical Context
Sarapin originates from Sarracenia purpurea, a North American plant referenced in medical and botanical literature throughout the twentieth century. These mentions reflect broader professional interest in naturally derived compounds and their exploration within licensed clinical environments.
Historical references to botanical injectables do not represent clinical claims; rather, they illustrate ongoing professional dialogue around plant based preparations.
Product Characteristics and Professional Distribution
Sarapin is supplied as a sterile aqueous preparation derived from Sarracenia purpurea. The formulation contains no steroids or narcotics and is distributed exclusively to licensed healthcare professionals for in office use. Manufacturers emphasize controlled production environments, lot traceability, and professional grade packaging to support consistent quality for clinical settings.
Contemporary Professional Context
In modern professional discussions, Sarapin is typically described by characteristics such as:
- Botanical origin
- Absence of steroids
- Non narcotic composition
- Availability restricted to licensed providers
These descriptors are informational only and do not imply therapeutic benefit. Decisions regarding the use of any injectable product remain solely within the judgment and legal scope of licensed practitioners.
Manufacturing and Quality Considerations
Healthcare professionals evaluating injectable products often review general factors related to manufacturing quality and supply chain reliability. These considerations apply broadly to injectable preparations and include:
- Controlled manufacturing standards to support sterility and consistency
- Documented ingredient sourcing, particularly for botanical extracts
- Lot traceability, including batch numbers and expiration details
- Professional only distribution policies to ensure appropriate handling
Such elements help practitioners assess whether a supplier aligns with expectations for clinical environments.
Interested in Sarapin? Discover detailed product information and clinical applications.
Learn More About SarapinProfessional Training and Use Environment
Injection based procedures require appropriate licensure, training, and adherence to local regulations. Healthcare professionals typically receive instruction through accredited programs, continuing education, or specialty specific coursework.
Educational materials in fields such as physical medicine, rehabilitation, and musculoskeletal care often emphasize:
- Sterile technique
- Proper preparation
- Regulatory compliance
- Documentation standards
These principles support responsible practice across a variety of clinical settings.
Regulatory and Ethical Considerations
In the United States, injectable products and clinical procedures fall under federal and state regulatory frameworks. Licensed healthcare professionals are responsible for ensuring that any products they obtain:
- Meet applicable regulatory requirements
- Are sourced through legitimate distribution channels
- Are used within their licensed scope of practice
Because of these requirements, professional discussions about Sarapin typically emphasize its background, manufacturing standards, and distribution policies. This approach keeps the focus on factual, product‑related information rather than clinical interpretation.
Conclusion
Sarapin is a botanical preparation that has appeared in professional discussions for many years and continues to be supplied to licensed healthcare providers. Understanding its origins, manufacturing considerations, and distribution policies can help practitioners evaluate suppliers and maintain high standards within their clinical environments. For general product information or professional inquiries, licensed providers may refer to the manufacturer’s resources: Healthcare professionals interested in injection procedures can also explore related information about trigger point injections here:
https://ussfgmp.com/trigger-point-injections.htmlProduct information
https://ussfgmp.com/sarapin.htmlProfessional inquiries
https://ussfgmp.com/sarapin-contact.htmlAs with all injectable products, Sarapin should only be obtained and used by licensed healthcare professionals operating within their scope of practice.
Tags: sarapin injection, sarapin injection cost, sarapin injection price, what is sarapin injection